The study of motile and non-motile cilia and flagella
Leishmania mexicana
Why do we study the flagellum?
Cilia and flagella are cellular projections built around a microtubule axoneme whose molecular architecture is highly conserved across eukaryotic groups. Cilia and flagella serve two main functions: motility and sensory. Many cell types (ranging from single-celled organisms such as Leishmania to mammalian sperm cells) use their motile flagellum for locomotion. Sensory cilia serve a wide range of functions. In the human body, for example, cilia are important for the detection of developmental signals, and the perception of chemical and mechanical stimuli. Single-celled organisms also use cilia and flagella for detection of signals from the environment. In motile flagella across diverse species and cell-types, two canonical waveforms are used in order to mediate fluid flow and cell locomotion: the symmetric and asymmetric waveform.
Leishmania, the model and the parasite.
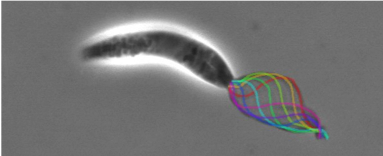
Leishmania have a single flagellum capable of generating symmetrical and asymmetrical waveforms. Their beating can be examined in live cells with videomicroscopy. The flagellar proteome has been studied (Beneke et al, 2019), the cells are genetically tractable, and there are high resolution cryo-EM maps of the microtubule doublets (Doran et al., 2025) and 96 nm repeat including the axonemal dyneins (Fochler et al., 2025). We are using this information to conduct mechanistic studies of structure-function relationships to understand how this complex organelle works.
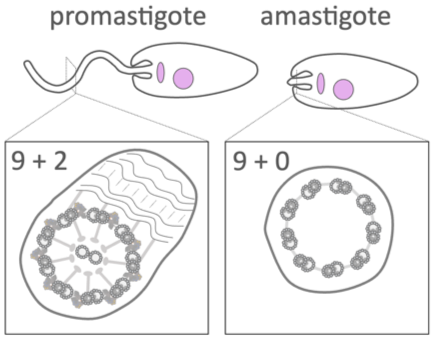
When the Leishmania parasite is engulfed by a macrophage, it changes shape and we discovered that its flagellum is remodelled from a device built for swimming (with a 9+2 arrangement of axoneme microtubules), into a structure resembling a sensory cilium (with a 9+0 axoneme) (Gluenz et al., 2010; Wheeler et al., 2015). We showed that motility of promastigotes is important for the colonisation of sand flies (Beneke et al., 2019) but not for the persistence in the mammalian host (Beneke et al., 2025).
How do we study the flagellum?
We have generated a Leishmania gene deletion library for >500 flagellar proteins, which we are using to study the functions of individual proteins in flagellar motility, to interrogate the dynamics of both waveform types in Leishmania, and to discover the function of cyclic nucleotide signalling in motility regulation.
Genetic tools: CRISPR-Cas9 gene deletion and mutagenesis, in situ tagging
Microscopy: Light and electron microscopy, ultrastructural expansion microscopy, high speed videomicroscopy for flagellar waveform classification and analysis of waveform parameters.
Assembly and Remodelling
Dyneins are large, multi-subunit motor complexes that require precise cytoplasmic pre-assembly before being transported to the axoneme. This process depends on a network of dynein assembly factors (DNAAFs), which can act as chaperones and scaffolding proteins to fold, stabilize, and assemble dynein subunits into functional complexes. We study the phenotypes of DNAAF knockout mutants to discover how they contribute to flagellar assembly and remodelling during the Leishmania life cycle.
Axonemal Dyneins
Axonemal dynein isoforms
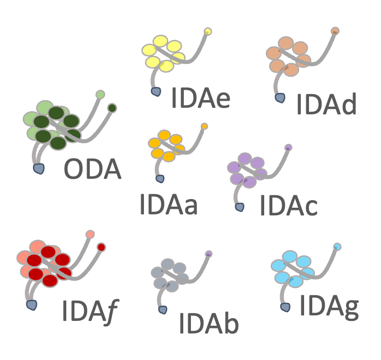
Dyneins are molecular motors which generate force. How the thousands of axonemal dyneins, and ten different isoforms, coordinate their activity to achieve complex biological behaviour, from the coordinated movements of multiciliary arrays to sperm chemotaxis and protist development, remains unclear. We use a range of tools to study the motilie cilium of Leishmania to explore
1) how each individual axonemal dynein isoform contributes to the two waveforms (Fochler et al., 2025)
2) how associated proteins regulate dynein activity.
Second Messengers
Eukaryotic motile cilia and flagella, from across diverse cell types and species, use second messengers like Ca2+ and cAMP to transduce signals and to modulate waveform dynamics. This leads to complex behaviours, often in response to environmental cues.
We explore how the signals are transduced to affect dynein driven beat mechanics.
Phosphorylation
Protein Kinase A is involved on the regulation of flagellar motility. We have shown that the catalytic subunit PKAC1 is indispensable: its absence causes a complete loss of the normal symmetric beat, leading to uncoordinated, flagellar beating (Fochler et al., 2023). Our current goal is to identify the specific axonemal protein targets of PKAC1. Defining this phosphorylation pathway will reveal how signals translate into coordinated motion. Such information could in time provide new insights insight into human ciliopathies like Primary Ciliary Dyskinesia (PCD).
Current Funding:
SNSF Project 320030L-227939